Weight Loss – Estrogen, Insulin and GLUT4 Receptors
Hormones play a huge role in blood sugar regulation and weight loss/gain. Estrogen especially as seen in PCOS (Polycystic Ovarian Syndrome), menopause, pregnancy and in men where testosterone levels start to drop off and estrogen increases due to a process called aromatization.
Understanding the role of estrogen and insulin in successful weight management is important as it affects so many people and is a constant frustration for especially women in why they can’t seem to lose weight with all the dietary changes and exercise that they do.
ESTROGEN AND THE HYPOTHALAMUS
The hypothalamus is the part of the brain that regulates food intake and appetite control and contains both ERα and ERβ receptors on the NPY (neuropeptide Y) neurons.
High levels of estrogen as seen during menstrual cycles and pregnancy may lead to decreased food intake and increased weight gain. Studies have found that when estrogen is removed, meal size and food intake tends to increase.
ESTRADIOL AND INSULIN
Estradiol (E2) is a beneficial type of estrogen.
When estradiol (E2) is balanced it improves insulin sensitivity and GLUT4 translocation, but when it’s either too high – such as during pregnancy, PCOS (Polycystic Ovarian Syndrome), abnormal menstrual cycles or obesity – or too low – such as during menopause – we may see a higher prevalence of Insulin Resistance (IR).
Estradiol in balance improves Insulin Resistance (IR), leptin sensitivity, the expression of insulin receptors on fat cells and also prevents fat accumulation.
These benefits become lost after menopause when estradiol levels drop off, but can also be reversed through estradiol hormone replacement which may then increase AMPK phosphorylation, expression of adiponectin and decrease expression of resistin. All of these contribute to weight loss.
Read more about estradiol and AMPK phosphorylation in these articles:
Peptides and Weight Loss – Focus on Leptin
How You Can Manipulate Peptides For Successful Weight Loss
Interestingly the exact opposite is true of men. Where men are concerned testosterone is the regulator of both insulin and leptin.
This is why estrogen is considered the youth hormone for women and testosterone the youth hormone for men.
Estradiol in women though improves blood sugar metabolism and reduces the risk for developing Insulin Resistance (IR) through the mechanism of encouraging glucose or sugar to enter muscle cells where they can be burned for energy, as long as you move your muscles (ie exercise).
So, when a woman’s estrogen levels fall as seen in menopause we may see blood sugar levels rise as well, which could trigger excess insulin release and contribute to weight gain. A common issue that women struggle with in their menopausal years.
ERα AND INSULIN
Why is ERα important?
Because low levels of ERα is considered a marker for obesity and increased risk for cardiovascular disease.
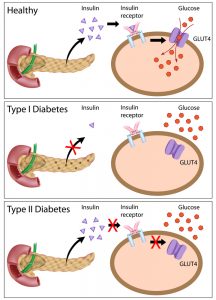
It is associated with Insulin Resistance (IR), increased leptin, increased cholesterol and smaller LDL particles as commonly seen with type 2 diabetes. ERα also regulates B cell proliferation in the pancreas and ER (Estrogen Receptor) stress is linked to type 1 diabetes with loss of B cell function.
NFkβ (Nuclear Factor Kappa-light-chain-enhancer) or activated B cells is a protein complex that controls cytokine production or inflammation.
NFkβ is triggered before and during B cell death. So we can see that there is a link between the loss of B cell function (or B cell death), inflammation, and its role in both type 1 and type 2 diabetes.
This article is not designed to go into the many things that can trigger inflammation but simply to demonstrate its role in disease and the development of metabolic syndrome.
ERα (ESTROGEN RECEPTOR) AND LIVER FUNCTION
Because Estrogen Receptors (ER’s) are involved in gene expression it goes without saying that disruption in these receptors will affect gene regulation in all the tissues they are found in.
In the liver low levels of ERα will increase expression of genes involved in glucose and cholesterol synthesis, and decrease expression of genes involved in cholesterol transportation.
What does this mean?
Well, it means that the liver will make more glucose and cholesterol – associated with metabolic syndrome, atherosclerosis and diabetes – and doesn’t carry it away.
The consequence being fatty liver.
And guess what?
There are also androgen receptors on the liver.
But to date no progesterone receptors can be found. This may be why a lot of women experience negative side-effects from oral contraceptives which usually contain estrogen and progestin. The synthetic progestin attaches to androgen receptors and have to be metabolised by the liver.
This puts an extra stress on the liver. But something else that can also add to this liver stress is ovulation.
The body makes extra estrogen after ovulation in preparation for pregnancy. If you are not pregnant then this extra estrogen has to be broken down in the liver. If the liver is stressed even more because of gluten intake, alcohol, medication use, or anything else, then estrogen levels will start to increase and bind to ER’s (Estrogen Receptors).
Estrogen is also a water-loving (hydrophilic) hormone, so cells responding to estrogen will tend to hold more water which contributes to symptoms resembling swelling such as headaches, breast tenderness and fluid retention.
CYP1B1 is a Phase I liver enzyme that is involved in breaking down estradiol into 4-hydroxy-estrone (4-OH) which can become toxic if it is allowed to build-up in the liver and escape, or gets reabsorbed from the digestive system due to increased beta-hyaluronidase activity. An upregulated CYP1B1 is associated with increased ERα and increased risk for endometriosis, fibroids and breast cancer. Methylation is the biochemical process that will neutralize these 4-OH’s into methoxy-estrone which is not harmful, which makes a well-functioning methylation cycle very important.
You can watch here what the methylation cycle is all about:
IMPORTANCE OF BLOOD SUGAR CONTROL
There are many nutrients needed for healthy blood sugar control which includes chromium, magnesium, alpha lipoic acid and many others. It is important to have healthy levels of these nutrients and good dietary intake during times of low estrogen.
 When blood sugar is poorly managed you may find that you need higher doses of estrogen than is ideal to feel ‘normal’
When blood sugar is poorly managed you may find that you need higher doses of estrogen than is ideal to feel ‘normal’
DHEA is another steroid hormone that can drop up to 80% between the ages of 25 – 75 years of age. When Insulin Resistance (IR) occurs there is decreased production of the adrenal hormone DHEA, the precursor to testosterone and estradiol, two hormones that are commonly low in post-menopausal women. Taking chromium not only reduces blood insulin and glucose, but also increases DHEA levels. This may lead to an increase in estradiol, and so provides an alternative treatment option for menopausal women by targeting Insulin Resistance (IR) in order to raise estrogen levels instead of giving them exogenous or artificial estrogen.
The same holds true for men. Low testosterone levels are found in men with diabetes or Insulin Resistance (IR).
INSULIN RESISTANCE AND HISTAMINE
Insulin upregulates mast cell degranulation and histamine release.
Hypoglycemia or low blood sugar (as seen when too much insulin is released) can activate H1 and H2 receptors (histamine receptors). Under normal circumstances low blood sugar will trigger the brain to release ACTH (Adreno Corticotropin Releaseing Hormone), which will trigger glucocorticoid production in the adrenals. Glucocorticoids inhibit the production of insulin in the pancreas so less glucose is carried via GLUT4 into the cells.
Abnormally high levels of histamine can interfere with this mechanism and fail to correct the hypoglycemia.
This increased activity in the HPA axis is often seen in type 1 and type 2 diabetics which may result in high cortisol levels. Theoretically this HPA dysregulation can be ‘fixed’ by increasing glucocorticoid receptor expression in the pituitary gland, inhibiting insulin release and thus histamine production.
This is important, because it means that anything that stimulates histamine either at very high or chronic levels may interfere with blood sugar regulation via too much insulin release and the ultimate development of Insulin Resistance (IR).
Similarly if Insulin Resistance (IR) improves either through supplementation or diet it may reduce histamine release and allergic reactions.
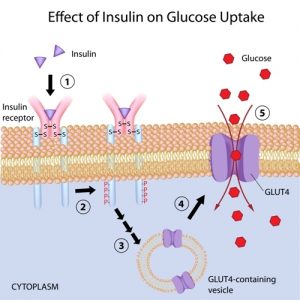
ESTROGEN AND GLUT4 RECEPTORS/TRANSPORTERS
GLUT4 is the glucose transporters (gates) on muscle, fat and liver cells that transport blood glucose into the cells for energy (ATP) production or energy storage (glycogen).
The opening of these gates or activation of these transporters are triggered by insulin. Insulin Resistance (IR) occurs when these transporters do not respond to insulin signalling. The body then secretes more insulin, but the sugar or glucose stays in the bloodstream because GLUT4 fails to listen and the gates do not open.
So how does estrogen influence this?
Estradiol (E2) can bind to two types of receptors and doesn’t seem to prefer one to the other:
- ERα (Estrogen Receptor α) signals for cellular proliferation and is linked to estrogen-cancers, Insulin Resistance (IR) and obesity. Estrone and raloxifene (selective estrogen receptor modulator used in breast cancer) will bind preferentially to this receptor.
- ERβ (Estrogen Receptor β) is anti-proliferative and will block the action of ERα. Estriol and genistein (from soy) will bind preferentially to this receptor. Androgens will also tend to decrease ERβ’s.
Once estradiol binds to one of these receptors they will move to the cell nucleus and DNA where they will regulate different genes.
ERβ does not seem to affect GLUT4 receptors in fat cells but ERα does.
Low ERα reduces GLUT4 expression. This will reduce glucose entry into the muscle cells, lead to Insulin Resistance (IR), high blood sugar levels, glucose intolerance and weight gain. Interestingly when estrogen is removed (as with a full hysterectomy) in those with low ERα, Insulin Resistance (IR) and weight loss improves.
ERβ and ERα combined in the same nuclei oppose each other’s actions and seem to have no effect on GLUT4.
Women with low ERα and low testosterone will be more likely to become Insulin Resistant (IR), have diabetes or gain weight. So if we increase ERα and testosterone levels we may improve the expression of GLUT4, reduce Insulin Resistance (IR) and lose weight more easily.
There are also nutrients we can use to help activate GLUT4 receptors and hold the key to weight loss.
What are these?
NUTRIENTS TO ACTIVATE GLUT4
- Chromium enhances GLUT4 expression on cell membranes.
- Alpha lipoic acid (ALA) enhances glucose uptake and GLUT1 and GLUT4 translocation to cell membranes especially if taken 30 minutes before a high carb meal.
- It seems Vanadium increases the express
As a side-note GLUT transporters are inhibited by lipid peroxidation. Lipid peroxidation occur when there are too many free radicals and not enough antioxidants, in other words too many toxins and not enough healthy foods.
INOSITOL
Inositol was one of the last nutrients we mentioned. Let’s elaborate on this a bit more.
There are two forms of inositol. Myo-inositol can be converted to D-chiro-inositol. They both seem to have glucose lowering effects but chiro-inositol works at much lower doses than myo-inositol. Type 2 diabetics seem to have higher urine excretion of both myo-inositol and chiro-insitol. Inositol does the same job as insulin, possibly through the phosphoinositide 3-kinase (PI3K) pathway, and will amplify the effect of insulin if they are both together, but it also spares insulin, allowing the body to release less when we eat a whole bunch of sugar, which takes pressure of the pancreas.
The type of inositol we use does become important though when we deal with fertility issues such as PCOS (Polycystic Ovarian Syndrome) even though they are both equally effective at glucose management (chiro-inositol just at much lower doses). Follicular fluid contains a ratio of myo-inositol:chiro-inositol at 100:1. It was found in studies that high doses of chiro-inositol led to poor egg quality whereas myo-inositol produced more mature eggs and more pregnancies.
So for overall weight loss chiro-inositol seems to be the more effective, but in younger women who are trying to conceive it is best to use the myo-inositol form.
THE LUTEAL PHASE AND INSULIN
The 2nd part of the menstrual cycle is called the Luteal phase where estrogen drops off and progesterone becomes the dominant hormone with the formation of the corpus luteum.
Diabetes becomes more resistant to treatment during the luteal phase, and also if cycles are irregular or too long – such as during a prolonged luteal phase. This is most likely due to lower estradiol (E2) levels.
Progesterone decreases the retention of ER’s (Estrogen Receptors) which causes a fall in estradiol (E2) levels. This is beneficial in conditions such as endometriosis where you have over-expression of ER’s (Estrogen Receptors) but maybe not within this context.
ESTROGEN AND PREGNANCY
During pregnancy women experience higher levels of estrogen. High concentrations of estrogen will tend to reduce GLUT4 expression and thus increase Insulin Resistance (IR) and the incidence of gestational diabetes.
SO, DO WE WANT TO STIMULATE ERα AND GLUT4?
Since ERα is linked to cancer and tumour growth we may need to look at this further.
The reason for this upregulation of GLUT4 via ERα is because cancerous cells need more glucose for growth, so they try to shunt as much glucose their way as possible. This is of particular importance in breast cancer as GLUT4 is expressed on fat cells. Hormone-dependent ER-positive breast cancer depend on estrogen for growth and proliferation for this reason and will push aromatization (the process through which testosterone is converted into estrogen) to increase estrogen levels.
When estrogen levels are low (ie not enough estrogen available to bind to upregulated Estrogen Receptors) due to the use of aromatase inhibitors for instance, the body will adapt as seen with tamoxifen-resistant breast cancer. Breast cancer cells become estrogen-resistant and in fact estrogen-independent, but ERα remain over-expressed and transcribe for genes that stimulate growth. In this case manipulating or decreasing estrogen may not be an effective strategy (xenoestrogens such as BPA would be the exception as described below). The focus may have to be on down-regulating ERα directly, not necessarily blocking ERα.
ERα is phosphorylated or activated by MAPK.
How You Can Manipulate Peptides for Successful Weight Loss
Phosphorylated ERα is a marker for prognosis in human breast cancer. Studies have found that vitamin D and dose-dependent estradiol (E2) down-regulated ER (Estrogen Receptors) by inhibiting phosphorylation of ERα.
It seems to be the over-exposure to estrogen (in particular xenoestrogens) and the over-expression of ER’s (Estrogen Receptors) that contribute to breast cancer. When these are in balance and xenoestrogens (or any other chemical with similar effects) are removed there doesn’t seem to be a problem.
The bottom line is that the aim is never to over-express or under-express anything but rather to modulate.
XENOESTROGENS
This is an interesting one.
Both estradiol and BPA (bisphenol A) increases insulin secretion and reduces blood glucose, however estradiol can still be blocked by ER blockers whereas studies suggest that BPA can not and have a rapid uncontrolled effect on ER’s. Similarly some metals such as cadmium and nickel can act as estrogen mimickers. They bind to ERα and take up the space that estradiol usually occupies on these receptors.
The body has an in-built control mechanism in how it regulates ER expression. In the presence of high estradiol (healthy estrogen) it will downregulate ER (through negative feedback) to keep gene transcription from going rogue. When xenoestrogens bind to ER’s instead it prevents estradiol from exerting this negative feedback mechanism that keeps everything in balance.
Since the breast tissue is made up of fat cells, this is where chemicals and xenoestrogens will accumulate. This could be one way in which metals, BPA and other xenoestrogens upregulate ERα, influence gene expression and transcribe for proliferative genes. This uncontrolled ERα upregulation result in similar uncontrolled upregulation of GLUT4 on these cells allowing an influx of glucose into cancerous cells to fuel their growth.
ANTI-OESTROGEN THERAPY AND INSULIN
When estrogen is decreased through anti-estrogen drugs such as tamoxifen or through removal of the ovaries, lipid metabolism becomes abnormal which increased the risk for Insulin Resistance (IR).
Will Improving Insulin Resistance Also Improve Estrogen Receptor Function?
Good question…
MENOPAUSE, HRT AND INSULIN RESISTANCE
During and after menopause estrogen levels drop.
This drop contributes to Insulin Resistance (IR) and metabolic syndrome. Hormone replacement therapy (HRT) tends to improve insulin sensitivity and glucose transportation into the cells. This is why most women feel better when they take synthetic or natural estrogen.
Deficiency in nutrients responsible for insulin regulation may increase the need or reliance on hormone therapy. When a woman needs to take high doses of hormones to make herself feel better it would be a good idea to look at blood sugar control. Some women report a feeling of ’emptiness’ in their breasts when they reduce their hormone replacement. This may be due to poor glucose entry into the fat cells of the breast and low glycogen stores. This may become a problem when women are taking abnormally high doses of estrogen in order to feel good which may not necessarily be healthy for them in the long run.
Improving the cell’s response to insulin through other means instead will allow them to balance their estrogen better without experiencing ‘withdrawal’ symptoms.
THE SIMPLIFIED VERSION
For those who are not necessarily interested in the biochemistry or need things to be a bit more simplified, I’ve attempted to explain it a bit differently:
When you eat sugar the pancreas respond by secreting insulin and insulin triggers GLUT4 to open the gates and glucose enter the cells. Insulin Resistance is when insulin comes knocking on the door and GLUT4 refuses to open it and let glucose in. Low insulin (as seen with type 1 diabetics) is when there is not enough insulin knocking on the door so GLUT4 doesn’t know it has to open it. In both these cases you have glucose sitting outside the cells waiting to be let in, but nobody is inviting them in. So they start to accumulate outside the door and this is when you see high blood glucose levels.
Now if the pancreas is able to it will respond by sending out more insulin to bang on the GLUT4 doors and demand glucose be let in. If GLUT4 still doesn’t listen you end up with lots of glucose and lots of insulin outside the door, as seen with high blood glucose and high blood insulin.
Certain nutrients (see Nutrients to Activate GLUT4 higher up) will force the GLUT4 doors open and let glucose in. As more glucose enters the cell, less is left hanging around outside the door so blood glucose levels drop. The pancreas also now sees that the doors are open so it sends out less insulin to go banging on the doors, and blood insulin levels start to drop.
Estrogen works similar to insulin. It binds to estrogen receptors which tells GLUT4 to open the doors. If there is enough estrogen doing this job and histamine is not abnormally high, the pancreas will not send so much insulin out to knock on GLUT4 doors. Similarly if your diet is healthy with limited sugar, good amounts of fats and proteins, and plenty of nutrients to support healthy blood sugar levels, the demand on estrogen will be less.
SO, THIS IS WHAT YOU NEED TO DO…
- Make sure your estrogen is balanced. If estradiol levels are low don’t try and detoxify estrogen even more. If estradiol is high maybe improve liver sulfuration and glucuronidation pathways.
- Make sure you are not exposed to any xenoestrogens or metals in your environment, and then detoxify whatever may be stored in the fat cells.
- If you have allergies or food intolerances, address these to get histamine under control.
References:
Muscle GLUT4 regulation by estrogen receptors ERβ and ERα
Insulin resistance of pregnancy involves estrogen-induced repression of muscle GLUT4
Vanadium increases GLUT4 in diabetic rat skeletal muscle
Activation of AMPK by bitter melon triterpenoids involves CaMKKβ
Estrogen Receptors and the Metabolic Network
ER Stress as a Trigger for β-Cell Dysfunction and Autoimmunity in Type 1 Diabetes
Interplay between insulin resistance and estrogen deficiency as co-activators in carcinogenesis.
Estrogen receptors and androgen receptors in the mammalian liver.







Hi, I was just wondering where progesterone fitted into insulin resistance, some doctors say if you use progesterone in menopause without estrogen it creates insulin resistance, and some websites say progesterone helps insulin resistance? Totally confused by this which is it?
Hi AP, it most likely depends on the level of progesterone. During pregnancy when progesterone is at its highest it may cause insulin resistance to allow more glucose for baby’s growth. During menopause estrogen drops off which may make you less insulin sensitive. I feel the ratios between the two hormones are probably more important here rather than looking at them individually, but it’s clear that certain live events such as pregnancy and menopause these ratios change which can then make it harder to lose weight.